Magnetism? Say What?

We did not believe the magnetism at first, but we kept seeing reports. So, the NZDSOS science team investigated. This is our report.
We have shared it with the authorities.
Magnetism in New Zealand Covid-19 Vaccinees: Evidence of Graphene Oxide or Contaminated Vials or Both?
Introduction
This article summarises concerns related to observations that Covid–19 vaccinees in New Zealand demonstrate magnetic properties over the site of their vaccination, what the possible biological mechanism of this phenomenon is, and why we believe this poses a threat to the health of New Zealanders who are undergoing vaccination. We believe the Ministry of Health should urgently investigate this issue since it poses an urgent clinical threat, particularly for patients who subsequently undergo magnetic resonance imaging. We also speculate that factors associated with this phenomenon may explain the some of the adverse events associated with Covid–19 vaccination. First, we draw attention to a biologically plausible mechanism for these effects.
The New Zealand government acquired Emergency Use Authorisation for the Pfizer–BioNTech Comirnaty mRNA COVID–19 vaccine against COVID–19 disease caused by SARS–CoV–2. The ingredient list for the Pfizer–BioNTech COVID–19 mRNA vaccine is listed within the FDA Comirnaty insert (1). They are:
- mRNA
- Lipids:[ALC–0315]:
- [ALC-0315]: (4– hydroxybutyl)azanediyl)bis(hexane–6,1–diyl)bis(2–hexyldecanoate)
- [ALC–0159]: 2 [(polyethylene glycol)–2000]–N,N–ditetradecylacetamide
- 1,2–Distearoyl–sn–glycero–3–phosphocholine
- cholesterol
- Potassium chloride
- Monobasic potassium phosphate
- Sodium chloride
- Dibasic sodium phosphate dihydrate
- sucrose.
There are two patented ingredients, namely ALC–0315 and ALC–0159, within the vaccine; hence information of these products are protected by intellectual property (IP) rules. Safety information about these are also redacted from publicly released documents. However, SINOPEG is the Chinese pharmaceutical and medical company that manufacture the PEGylated lipid nanoparticles in the Pfizer and Moderna vaccines. There are lipids used that are currently under patent (protected by IP law):
- Moderna: [SM–102] (https://www.sinopeg.com/heptadecan–9–yl–8–2–hydroxyethyl–6–oxo–6–undecyloxy–hexyl–amino–octanoate–sm–102–cas–2089251–47–6_p480.html)
- Pfizer–BioNTech: PEGylated lipid2 [ALC–0159] (https://www.sinopeg.com/2–polyethylene–glycol–2000–n–n–ditetradecylacetamide–alc–0159–cas–1849616–42–7_p477.html)
- Pfizer–BioNTech: Cationic lipid2 [ALC–0315] (https://www.sinopeg.com/4–hydroxybutyl–azanediyl–bis–hexane–6–1–diyl–bis–2–hexyldecanoate–alc–0315–cas–2036272–55–4_p476.html).
Both ALC–0159 and ALC–0315 safety data is also hidden from the contact hazards section for Comirnaty completely (1); however, there is evidence of graphene oxide being used by SINOPEG in the development of their PEGylated lipid nanoparticles (3). Additionally, there is a patent held by Shanghai National Engineering Research Center for Nanotechnology Co Ltd, for worldwide applications titled ‘Nano coronavirus recombinant vaccine taking graphene oxide as carrier’ (Patent number: CN112220919A; Date: 2020-09-27; Country: China), clearly showing graphene oxide is being used in COVID-19 vaccine development (which company is undetermined).
The World Health Organization (WHO) assessment on the Pfizer-BioNTech vaccine, specifically the SINOPEG PEGylated lipid nanoparticles state that “The primary function of the PEGylated lipid ALC-0159 is to form a protective hydrophilic layer that sterically stabilises the lipid nanoparticle, which contributes to storage stability and reduces nonspecific binding to proteins.” (4) It is then discussed that ALC-1059 may result in anaphylactic reactions (5) and refers to the need for analysis by NIAID and the FDA to examine this in “people with high levels of anti-PEG antibodies or have experienced severe allergic responses to drugs or vaccines before.” (4)
A lack of adequate safety data for ALC-0159 and ALC-0315 is repeated in various reports: MEDSAFE (6), European Medicines Agency (EMA) (2), WHO and even Pfizer’s own safety data sheets (7); and some organisations have requested more information of the safety of the use of these and a breakdown of the production process of this chemical.
In New Zealand, MEDSAFE has granted provisional consent for use of the product until 3rd November 2021 and this use was subject to a number of conditions. (6) It requires that the New Zealand Sponsor must fulfil the following obligations within the timelines specified, the dates of which may be altered by mutual agreement with MEDSAFE. Most of these conditions needed to be assessed by the 9th of August. Of the 58 listed conditions, 20 relate to the two ingredients ALC-0159 and ALC-0315 relating to the breakdown of materials, their production and safety data.
Since these are novel excipients (used for the first time) then why have they had no pharmacokinetic studies been done on them?
This question was also queried by Peter Roderick in BMJ (Rapid Response: BMJ 2021;372:n627) (8): “I also see that the EMA’s Public Assessment Report [4] states that in January 2021 (and April 2021) “additional information about the synthetic process and control strategy for” ALC–0315 and ALC–0159 “should” be provided by BioNTech, with final reports in July 2021, in order to “confirm the purity profile and ensure comprehensive quality control and batch–to–batch consistency throughout the lifecycle of the finished product””.
Magnetofection
Magnetofection is a process where gene vectors are associated with superparamagnetic nanoparticles to result in targeted gene delivery through an application of a magnetic field9,10, and is currently used in biotechnology (11) and biomedicine (11) in cancer treatments (11), drug delivery (11,12), vaccine delivery (13,14) and cellular imaging (11). In the case of drug and vaccine delivery, magnetofection utilises the properties of magnets, such as graphene oxide (GO) (15,16) (an oxidised derivative of graphene) (11) or iron oxide nanoparticles (IONPs) (17) (or superparamagnetic iron oxide nanoparticle, SPIONs) (18), to infect the cell and deliver genetic material (DNA or RNA) into the cell with greater efficacy. However, evidence shows that GO and GO nanoparticles have a toxic effect on living cells and organs (11,19), and pose a risk to individuals exposed (19). It has been speculated that the interactions between graphene oxide with blood proteins and biological membranes could lead to severe effects, such as thrombogenicity and immune cell activation (20). Superparamagnetic iron oxide nanoparticle, SPIONs, may be toxic, so the iron oxide core is coated by an organic or inorganic layer.21 A depiction of this process is shown in Figure 1, obtained from Chandra and colleagues (2014) (22) shows the use of magnetic materials in magnetofection to enable genetic material to enter into the cell.
Figure 1
Figure 1. Schematic representation of the process of magnetofection by magnetic dendriplexes. (22)
There are serious concerns of the impact of GO in the body. Researchers identified that coating GO in lipid bilayers results in low toxicity (23), and greatly decreases the haemolytic properties (24). The article states “GO also interacts with red blood cells and causes hemolysis; hemolysis is decreased when GO is previously coated with lipid membranes, particularly with pure phosphatidylcholine vesicles.” [Emphasis added.] These articles do not conclude that toxicity or haemolysis is eliminated. Ryan Cross reports in his article that Without these lipid shells, there would be no mRNA vaccines for COVID–19 25 which states “Fragile mRNA molecules used in COVID–19 vaccines can’t get into cells on their own. They owe their success to lipid nanoparticles that took decades to refine.” The story then goes on to discuss the fragile nature of mRNA and the protection afforded by these lipids. A review by Schoenmaker and colleagues (2021) (26) highlights that “current COVID–19 mRNA–LNPs vaccines must be stored at (ultra)low temperatures.” The EMA additionally states stability of ALC–0315 and ALC–0159 when stored at the recommended storage conditions.(2)
In addition to this, the EMA also acknowledges that “Lipid related impurities have been observed in some recently manufactured finished product batches, correlated with ALC–0315 lipid batches. The quality of ALC–0315 excipient is considered acceptable based on the available data on condition that specific impurities in the finished product will be further evaluated.”( 2) The purity profile is also yet to be confirmed. These admissions of ALC–0315 impurities are of concern. If ALC–0315 is used to coat GO (or SPIONs) nanoparticles, this could result in free–GO or SPION nanoparticles circulating and have harmful effects on the body. If storage conditions are not met, what impact do these have on the integrity of these lipids covering the mangetofection agent?
Is there evidence of graphene oxide in the Pfizer–BioNTech “Comirnaty” mRNA vaccination?
Is GO used in the production of the Pfizer–BioNTech mRNA COVID–19 vaccination? Due to the presence of two patented ingredients being used in the Pfizer–BioNTech COVID–19 mRNA vaccine, the use of GO cannot be definitively determined. However, outside evidence suggests that it is.
Ex–Pfizer Employee
Karen Kingston, an ex–employee at Pfizer has stated that Pfizer is using graphene oxide to encapsulate their pegylated lipid nanoparticle and that the composition of this is present in their patent. (27)
Chemical Analyses of COVID–19 Vaccines
Spanish scientists found that toxic levels of graphene oxide were found in a vial of Pfizer/BioNTech vaccine, using spectroscopy and electron microscope analysis. (28) As reported:
“Graphene oxide was identified in samples from all the major Big Pharma players, including AstraZeneca, Pfizer, Moderna, Sinovac, Janssen, and Johnson & Johnson. Certain COVID shot vials contained as much as 99% graphene oxide and not much else….
With so much concern surrounding this toxic compound, why is it being foisted upon the public en masse?
According to its safety data sheet, there is “no data available” on the various toxicological effects of graphene oxide, including acute toxicity, carcinogenicity, reproductive toxicity, and skin and eye damage. However, 2012 research published in the peer–reviewed journal ACS Nano concludes that inhaling these nanoparticles can cause lung inflammation and lead to lung cancer and pneumonia.
The Spanish researchers who discovered graphene oxide in COVID shots also note that this toxic compound can also:
- Promote thrombus formation (blood clots)
- Damage red blood cells
- Damage the immune system Inflame mucous membranes and contribute to a loss of taste or smell – or even lead to an unusual metallic taste in the mouth, which has been reported
- Exert magnetic properties once inside an organism – which may explain the bizarre footage of people holding magnetic objects to their arms following their jabs.”
The Scientist Club released a report title Nanotechnological investigations on Covid–19 vaccines where they used chemical analyses to determine the chemical make up of vaccines. In addition to the liposomes expected, the main findings determined that there was evidence of graphene oxide in the Pfizer vial through:
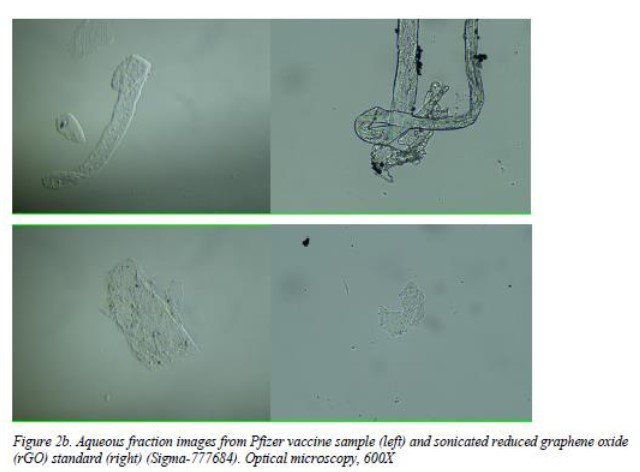
- Optical microscopy: showing corrugated and flat, irregular transparent sheets of variable size and shape.
- Scanning (SEM) and Transmission (TEM) Electron Microscope observations: presence of graphene is confirmed.
- UV absorbance and fluorescence spectroscope: evidence of graphene–like sheets that were abundant in suspension in the sample; peak values of fluorescence in accordance with peak values for GO.
Figure 1

Figure 2a
Figure 2b
The Scientists’ Club report additionally found the following contaminants (debris) in COVID–19 vaccine vials, using Environmental Scanning Electron Microscope coupled with an x–ray microphone of an Energy Dispersive System:
| COVID-19 VACCINE | Contaminants Identified (Debris) |
| Pfizer | • Graphene oxide. • Sharp debris of 20micron: made up of carbon, oxygen chromium, sulphur, aluminium, chloride, nitrogen. • White 2–micron particle: bismuth, carbon, oxygen, aluminium, sodium, copper, nitrogen. • Organic (carbon–oxygen–nitrogen) aggregate embedded with nanoparticles of bismuth–titanium–vanadium–iron–copper silicon–aluminium. |
| Moderna | • Graphene oxide. • Mixed entity (organic–inorganic): carbon–based substrate with nanoparticles embedded. Nanoparticles composed of aluminium–copper–iron–chlorine. • Silicon–lead–cadmium–selenium (highly toxic combination reminds researchers of quantum dots: cadmium selenide). • 100–micron entity, reminding of graphene, composed of carbon and oxygen with contamination of nitrogen, silicon, phosphorus, chlorine. • Carbon–based entities mixed with aggregates filled with aluminium–silicate particles. |
| Astra Zeneca | • Stainless steel nanoparticles: iron–chromium–nickel. |
| Janssen | • Stainless steel, glued together with carbon–based glue. |
Table 1. A list of contaminants in COVID–19 vaccine vials identified by The Scientists’ Club.
The Scientists’ Club conclude with the following Discussion:
The analyzed “vaccines” present components that are not mentioned in the technical data sheet and whose presence does not seem to have to do with the concept of vaccine. Since they are not included in the documentation presented to the Governmental organizations (FDA, EMA, etc.) for the legal approval aimed at the commercialization and the human use, they seem to be a contamination probably due to the industrial process of manufacturing. It seems that nobody controlled the final product before its distribution. That means that consumers are not informed of the real content of the products. Possible side effects may be due to the injection of those contaminants into the body. It must be observed that the components that are not declared but we identified are not biocompatible and some have also a mechanical impact once they are inside the blood circulation, especially in contact with the vascular endothelium.
The entities present in Pfizer and AstraZeneca “vaccines”, identified by the ESEM images, can represent a risk for the human body. They can be responsible of the formation of thrombi, since they are thrombogenic. A further risk is represented by the extravasation of the particles with an ensuing possible haemorrhage. Once in the blood circulation, the particles can be carried also to the brain. In this case the patient can suffer from a stroke, and/or a cerebral haemorrhage. If the damage of the endothelium caused by the particles occurs in the heart, there is a high probability of contracting a myocarditis. In addition to all that, the toxicity of graphene is well–known.
The presence of non–biocompatible organic–inorganic foreign bodies in the blood circulation can be responsible of a nano–bio–interaction that can induce severe health problems.” [Emphasis added]
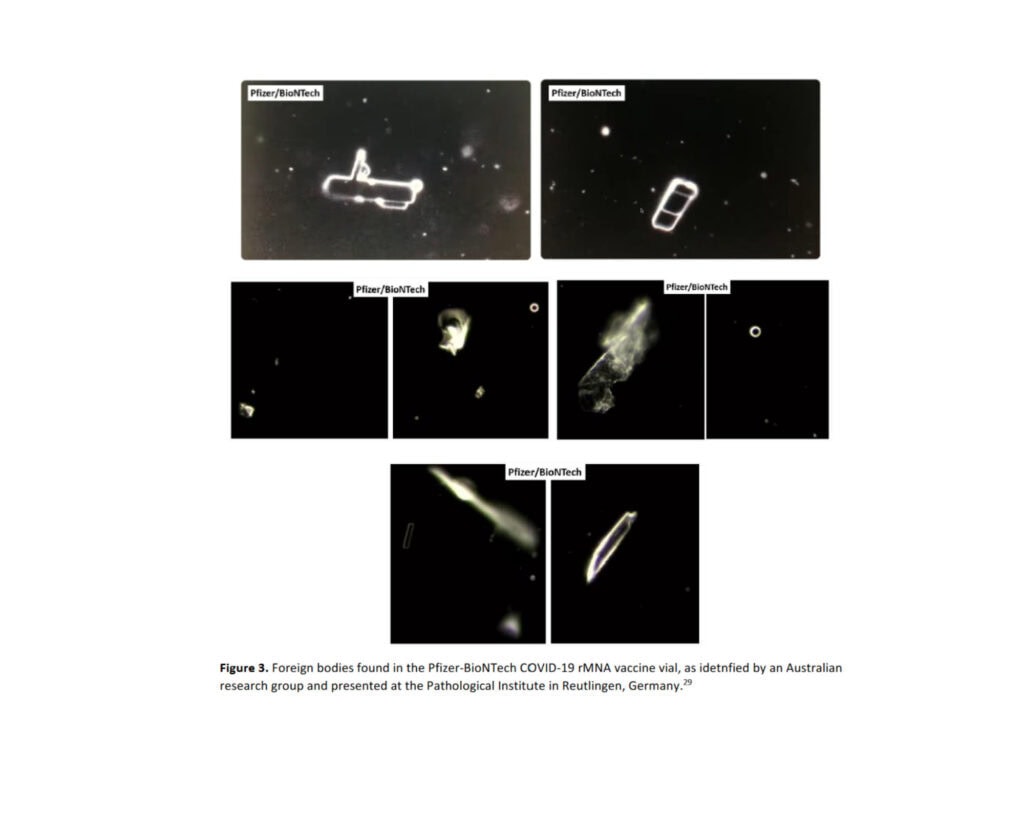
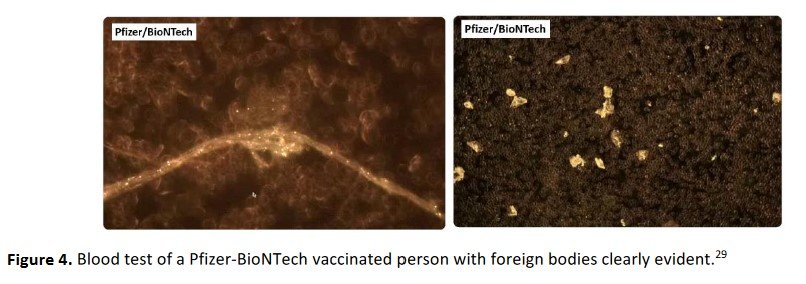
A press conference (29) at the Pathological Institute in Reutlingen, Germany presented findings from an Austrian research group investigating the vaccines. They, too, identified foreign bodies in the Pfizer–BioNTech COVID–19 vaccines. Images of foreign bodies are shown below.
Figure 3
Figure 4
Contamination
If we cannot determine whether GO is intentionally used in the pharmaceutical design of their two patented ingredients, for the purpose of magnetofection, as reported by an ex-Pfizer employee, found present in vials by
Spanish scientists and The Scientists’ Club; could the magnetism being experienced be caused by contamination of foreign bodies, as identified by The Scientist Club and Austrian researchers? This raises the question of serious vaccine manufacturing quality and control issues.
Japan
The use of Moderna and Pfizer-BioNTech COVID-19 vaccines in Japan has revealed contaminants on three separate occasions.
- On 26 August 2021, it was reported that 1.6M doses of Moderna were removed from use after contamination by a substance that reacts to magnets was round (30).1.6M doses of Moderna were immediately recalled and investigations followed, determining that it was stainless steel (31) from a manufacturing site in Spain. This contamination was initially reported not to pose an issue to the recipient’s health through statements released from the government, as well as Takeda Pharmaceutical and Moderna in Japan, but later reported to have resulted in three deaths.
- Within a week of the stainless-steel contamination, another Moderna vial was found to have black floating particles, resulting in approximately 560,000 Moderna vials being recalled (31).
- Following these first two events, Japan again reported contamination of Pfizer COVID-19 vials identified to have white floating matter at multiple sites in Sagamihara, one site in Kamakura and one site in Sakai (32). The vials were from batch number FF5537, and were immediately recalled. The Scientists’ Club also identified white debris (contamination) in their samples, which they analysed to be made of bismuth, carbon, oxygen, aluminium, sodium, copper, nitrogen.
The death of three people reported to date in Japan linked to receiving Moderna vaccines containing stainless–steel contaminants (31) would suggest that contaminants reacting to magnets are of concern and cannot be ignored, as suggested by the joint statement between Takeda Pharmaceutical and Moderna in Japan (30). This additionally calls into question the integrity of the pharmaceutical companies to ensure safe manufacturing practices, quality control, and taking responsibility of poor–quality vaccines with increased manufacturing and supply pressures.
New Zealand
The use of Pfizer–BioNTech Comirnaty mRNA vaccines have been rolled out in New Zealand and are considered safe for use in those 12 years of age and up. However, the recent emergence of videos online of recipients experiencing magnetism associated with recent sites of vaccination is of great concern. There is no physiological basis for magnetism in the body. Whilst all life relies on the flow of tiny currents which produces electromagnetic fields related to Faraday’s law, palpable magnetism of this nature is unprecedented and does not exist in nature. Here, we outline these concerns and present a preliminary study of reported magnetism in New Zealand during the period Thursday, 16th September until Thursday, 23rd September 2021, inclusive.
Method
Following reports of magnetism on Facebook and other social media platforms, individuals experiencing this phenomenon were contacted and data was collected (researcher holds appropriate postgraduate degrees in
science). This was to determine the extent of magnetism in New Zealand, and whether there was evidence of batch numbers of concern, such as observed in Japan; and whether it was localised to one part of New Zealand or more widespread.
Evidence of magnetism was obtained, where possible, in the form of the submission of videos or photos or from personal experience observing the magnetism; as well as a copy of their vaccination card (where possible). Location and batch numbers for each vaccination (first and/or second) were recorded (when available). Simple demographic characteristics such as gender, ethnicity and location were also reported, where possible.
Assistance is being provided to those wishing to submit a CARM report, following reports that the Ministry of Health did not further investigate claimants who reported this evidence, which when reported back to the group forums, discouraged others from reporting their experiences.
Results
There were 41 reports of magnetism during the period Thursday, 16th September to Thursday, 23rd September 2021, in New Zealand; proof of magnetism and vaccination was provided for 14 individuals; with an additional two providing partial evidence (video, but no vaccination card). Of these 14 with proof of vaccination, seven received one dose and seven received two. Partial or full batch numbers were missing on vaccination cards for six vaccine recipients (not filled out by vaccinator or no space available on their vaccination card for the batch number to be recorded). There were 12 reports of magnetism in males, 22 in females, and in 7 cases the gender was unknown.
It was common to additionally be provided information and discussion on adverse events additional to the magnetism that subjects experienced. These included, but were not limited to: chest pain, blood pressure issues, dizziness, headaches (severe), electric shocks from mobile phones, electrosensitivity, and twitching muscles. One sufferer had to be admitted to hospital with serious symptoms. A number of sufferers have requested assistance in submitting CARM reports (underway) and medical advice (we referred them to their GP). Some reported that they approached the New Zealand Ministry of Health or GPs to get answers and assistance reporting to CARM; and were dismissed, discouraging from other’s from reporting their experiences as they felt it wasn’t worth their time. They have additionally been mocked and ridiculed on national TV, social media, and sadly, by family members and friends. Of note, and concern, is that reporting has been disproportionately from Maori and Pacific Island populations, but we recognise this could be due to the sampling method and/or the focused rollout of the vaccination program in Auckland during their drawn-out lockdown, including the reported targeted vaccination programs of these ethnic groups.
Distribution of magnetism in New Zealand
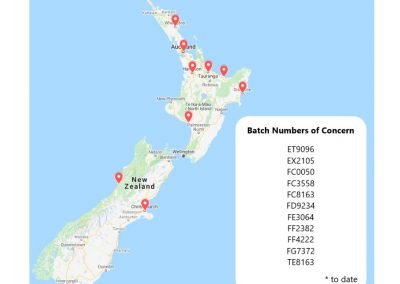
A map is provided of the location of vaccinations administered in New Zealand of those experiencing magnetism, and shows that magnetism is widespread, especially throughout the North Island and the top half of the South Island. Those experiencing magnetism supplied evidence of receiving one of 11 batch numbers associated with magnetism, and are as follows: ET9096, EX2105, FC0050, FC3558, FC8163, FD9234, FE3064, FF2382, FF4222, FG7372, TE8163. Of particular note and concern, are those starting with FF#### (as found in Japan’s Pfizer contaminated batch, FF5357).
Figure 5
There have been > 40 reports of magnetism located throughout New Zealand, in 9 locations. Batch Numbers of concern form those who submitted evidence of magnetism and vaccination batch number, to date.
Discussion
Magnetism has been reported in 41 individuals in New Zealand temporally associated with COVID–19 vaccination. There were 11 batch numbers associated with magnetism, of particular note and concern, are those starting with FF#### (as found in Japan’s Pfizer contaminated batch, FF5357). Given the New Zealand government acquired 250,000 vials of Pfizer–BioNTech vaccines from Spain on 9th September 2021 33, with no Spanish manufacturing, product testing or packaging company listed as those authorised by MEDSAFE6; we hold great concern over the quality of these vials given Japan’s experience with Moderna contamination from Spanish manufacturing sites: (i) stainless steel resulting in magnetism (reported 26th August 2021); and (ii) black floating objects; and following this, (iii) Pfizer vials with white floating objects (reported 15th September 2021).
As you saw in the evidence provided above,
- Graphene oxide has magnetic properties.
- Graphene oxide has been used in lipid nanoparticle delivery of drugs (magnetofection).
- Graphene oxide is extremely harmful to the body. The lipid covering decreases toxicity to the body somewhat, but does not eliminate it.
- Graphene oxide has been found to be contained within the Pfizer–BioNTech mRNA COVID–19 vaccine through chemical analysis of vials from two separate research groups, and reported by an ex–Pfizer employee.
- The lipid nanoparticle vaccines are unstable and require ultra–low temperatures.
- The EMA highlighted issues with lipid related impurities being observed.
- There is evidence of contamination issues with the Pfizer–BioNTech mRNA COVID–19 vaccine, as identified by The Scientists’ Club and Austrian researchers, and witnessed in another vaccine in Japan (Moderna).
- New Zealanders are experiencing magnetism as a result of vaccination from at least 11 batches of Pfizer–BioNTech Comirnaty mRNA COVID–19 vaccines, distributed at least throughout the North and top half of the South Island.
Is this magnetism we are experiencing evidence for:
Contamination (a serious quality control issue)
AND/OR
the result of Graphene oxide (GO) escaping the lipid coating that should protect the body from the harmful effects of GO, and therefore, a substandard vaccine with impurities
(manufacturing issue) or has degraded (incorrect storage conditions),
AND/OR
the vaccine is unstable and unpredictable in its design (Pharmaceutical formulation issue) resulting in free, uncoated GO circulating the body?
Serious Health Implications
If the magnetism being experienced in New Zealand is because of one of the above, there are serious health implications of these contaminants, as described above, including:
Graphene Oxide:
- Promote thrombus formation (blood clots)
- Damage red blood cells
- Damage the immune system
- Inflame mucous membranes and contribute to a loss of taste or smell, or metallic taste in the mouth
Exert magnetic properties once inside an organism, which may cause problems particularly in patients who undergo magnetic resonance imaging.
Metal contaminants:
- Formation of thrombi (since they are thrombogenic) Haemorrhages
- Carried to the brain (once in bloodstream): stroke and/or cerebral haemorrhage
- Myocarditis.
Induction of severe health problems.
Given the significant health implications identified in Japan from contaminants that react to magnets, namely, the death of three recipients; we urge medical practitioners, the Ministry of Health helpline and any others approached by someone experiencing this phenomenon to take their complaint and concerns seriously. Historically, such complaints would have been met with an appropriate level of duty of care, given the underlying seriousness of such allegations. Unfortunately, during the roll–out of the New Zealand COVID–19 vaccination program, recipients of these complaints have not received a response to CARM reports submitted to date; experienced a complete lack of acknowledgement or concern shown to callers of the Ministry of Health helpline; and been dismissed on national television by medical practitioners and scientists (encouraged by the media).
Conclusions
The evidence and results presented in this report present alarming findings. Of the 41 reported cases of magnetism (to date), alongside concerning side effects post–vaccination reported, at least 11 batches of Pfizer–BioNTech COVID–19 mRNA vaccines distributed in at least 9 cities throughout New Zealand were found to be temporally associated with magnetism. These findings point to the possible contamination of vials of Pfizer–BioNTech Comirnaty COVID–19 mRNA vaccine vials in New Zealand, or toxicity from graphene oxide, which must be investigated thoroughly. The experience of magnetism causes great distress and we believe should be investigated transparently due to the association with serious health conditions from the presence of graphene oxide and other contaminants.
We call for an immediate halt on any vials with the batch number ET9096, EX2105, FC0050, FC3558, FC8163, FD9234, FE3064, FF2382, FF4222, FG7372, TE8163; in addition to any others purchased from manufacturing sites in Spain, until independent analyses can be carried out to ensure the quality of these batches, manufacturing lines and production sites to check for the presence of GO or contamination by other debris.
References
1. Vaccine information fact sheet for recipients and caregivers about Comirnaty (COVID-19 vaccine, mRNA) and Pfizer-BioNTech COVID-19 vaccine to prevent coronavirus disease 2019 (COVID-19). Available from:
https://www.fda.gov/media/144414/download
2. European Medicines Agency. Assessment report: Comirnaty. 2021 February 19. Available from: https://www.ema.europa.eu/en/documents/assessment-report/comirnaty-epar-public-assessment-report_en.pdf
3. SINOPEG. Industry News: Core-shell structured polyethylene glycol functionalized graphene for energy-storage polymer dielectrics: Combined mechanical and dielectric performances. Written 2020 Sept 1. https://www.sinopeg.com/core-shell-structured-polyethylene-glycol-functionalized-graphene-for-energy-storage-polymer-dielectrics-combined-mechanical-and-dielectric-performances_n28
4. World Health Organization. Evidence Assessment: Pfizer-BioNTech COVID-19 vaccine. 2021 Jan. Available from: https://www.who.int/docs/default-source/immunization/sage/2021/january/4-evidence-assessment5-jan-2021-final.pdf?sfvrsn=cf627b70_9
5. Cabanillas B, Akdis, CA, Novak N. Allergic reactions to the first COVID-19 vaccine: A potential role of polyethylene glycol? Allergy. 2021;76:1617–1618. DOI: 10.1111/all.14711
6. MEDSAFE. Medsafe Product Details: Comirnaty (COVID-19 mRNA vaccine). [Cited 2021 Sept 26] Available from: https://www.medsafe.govt.nz/regulatory/ProductDetail.asp?ID=21938
7. Vaccine information fact sheet for recipients and caregivers about Comirnaty (COVID-19 vaccine, mRNA) and Pfizer-BioNTech COVID-19 vaccine to prevent coronavirus disease 2019 (COVID-19). Available from:
https://www.fda.gov/media/144414/download
8. Peter E. Roderick. Rapid Response to: The EMA covid-19 data leak, and what it tells us about mRNA instability. BMJ 2021; 372 doi: https://doi.org/10.1136/bmj.n627 (Published 10 March 2021) Available from:
https://www.bmj.com/content/372/bmj.n627/rr-1
9. Scherer, F., Anton, M., Schillinger, U. et al. Magnetofection: enhancing and targeting gene delivery by magnetic force in vitro and in vivo. Gene Ther 9, 102–109 (2002). https://doi.org/10.1038/sj.gt.3301624
10. Magnetofection. Chemicell. Available from: http://www.chemicell.com/products/Magnetofection/Magnetofection_separation.html
11. Rhazouani A, Gamrani H, El Achaby M. et al. Synthesis and Toxicity of Graphene Oxide Nanoparticles: A Literature Review of In Vitro and In Vivo Studies. Hindawi BioMed Research International. 2021. https://doi.org/10.1155/2021/5518999
12. Czugala M, Mykhaylyk O, Böhler P, Onderka J, Stork B, Wesselborg S, Kruse FE, Plank C, Singer BB, Fuchsluger TA. Efficient and safe gene delivery to human corneal endothelium using magnetic nanoparticles. Nanomedicine (Lond).
13. Xu L, Xiang J, Liu Y, Xu J, Luo Y, Feng L, Liu Z, Peng R. Functionalized graphene oxide serves as a novel vaccine nano-adjuvant for robust stimulation of cellular immunity. Nanoscale. 2016 Jul;11(14):1787-800. doi: 10.2217/nnm-2016-0144. Epub 2016 Jul 7. PMID: 27388974.
14. Wang X, Cao F, Yan M, et al. Alum-functionalized graphene oxide nanocomplexes for effective anticancer vaccination. Acta Biomaterialia. 2019; 83: 390-399.
15. He Y, Yi C, Zhang X, Zhao W, Yu D. Magnetic graphene oxide: Synthesis approaches, physicochemical characteristics, and biomedical applications. TrAC Trends in Analytical Chemistry. 2021 Mar; 136. https://doi.org/10.1016/j.trac.2021.116191
16. Sarkar SK, Raul KK, Pradhan SS, Basu S, A. Nayak. Magnetic properties of graphite oxide and reduced grapheneoxide. Physica E: Low-dimensional Systems andNanostructures. 2014; 64: 78/82. https://doi.org/10.1016/j.physe.2014.07.014
17. Crespo-Barreda A, Encabo-Berzosa MM, González-Pastor R, Ortíz-Teb P, Iglesias M, L.Serrano J, Martin-Duque P. Chapter 11 – Viral and Nonviral Vectors for In Vivo and Ex Vivo Gene Therapies. Translating Regenerative Medicine to the Clinic. 2016: 155-177. https://doi.org/10.1016/B978-0-12-800548-4.00011-5
18. Mok H, Zhang M. Superparamagnetic iron oxide nanoparticle-based delivery systems for biotherapeutics. Expert Opin Drug Deliv. 2013 Jan;10(1):73-87. doi: 10.1517/17425247.2013.747507. Epub 2012 Dec 1. PMID: 23199200; PMCID: PMC4167583
19. Lategan K, Alghad H, Bayati M, Fidalgo de Cortalezzi M, Pool E. Effects of Graphene Oxide Nanoparticles on the Immune System Biomarkers Produced by RAW264.7 and Human Whole Blood Cell Cultures. Nanomaterials. 2018; 8, 125. doi:10.3390/nano8020125
20. Palmieri V, Perini G, De Spirito M, Papi M. Graphene oxide touches blood: in vivo interactions of bio-coronated 2D materials. Nanoscale Horizons. 2019: 2
21. Ghaffari M, Moztarzadeh F, Mollazadeh-Bajestani S. Chapter 3 – Drug delivery nanosystems for musculoskeletal regeneration. Nanoengineering in Musculoskeletal Regeneration. 2020; 77-103. https://doi.org/10.1016/B978-0-12-820262-3.00003-7
22. Chandra S, Nigam S, Bahadur D. (2014). Combining Unique Properties of Dendrimers and Magnetic Nanoparticles Towards Cancer Theranostics. Journal of biomedical nanotechnology. 10. 32-49. 10.1166/jbn.2014.1698.
23. Xu L, Xiang J, Liu Y, et al. Functionalized graphene oxide serves as a novel vaccine nano-adjuvant for robust stimulation of cellular immunity. Nanoscale. 2016 Feb 14;8(6):3785-95. doi: 10.1039/c5nr09208f. Epub 2016 Jan 27. PMID: 26814441.
24. Monasterio BG, Alonso B, Sot J, et al. Coating Graphene Oxide with Lipid Bilayers Greatly Decreases Its Hemolytic Properties. Langmuir 2017, 33, 33, 8181–8191. https://doi.org/10.1021/acs.langmuir.7b01552
25. Cross R. Without these lipid shells, there would be no mRNA vaccines for COVID-19. March 6, 2021. https://cen.acs.org/pharmaceuticals/drug-delivery/Without-lipid-shells-mRNA-vaccines/99/i8
26. Schoenmaker L, Witzigmann D, Kulkarni JA, et al. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. International Journal of Pharmaceutics. 2021 May 15; 601: 120586. https://doi.org/10.1016/j.ijpharm.2021.120586
27. Love A. CONFIRMED! Graphene oxide main ingredient in Covid shots. 2021 Aug 9. Available from: https://ambassadorlove.wordpress.com/2021/08/09/confirmed-graphene-oxide-main-ingredient-in-covid-shots/
28. Middleton S. Urgent: Toxic substance found inside Pfizer COVID shot, according to Spanish researchers. [Cited: Sept 20, 2021]. Available from: https://www.naturalhealth365.com/mrna-jabs-content-3898.html
29. Dr John. B. [cited 2021 Sept 21] Available from: https://threadreaderapp.com/thread/1440093324472459265.html
30. Menahan C. Japan: 1.6M Moderna doses pulled after officials discover vials contained ‘foreign matter’ that ‘reacts to magnets’. [Cited: Sept 20, 2021] Available from: https://www.sgtreport.com/2021/08/japan-1-6m-moderna-doses-pulled-after-officials-discover-vials-contained-foreign-matter-that-reacts-to-magnets/
31. Qatar Day. Japan finds another Moderna vial suspected to contain foreign substance. 2021 Sept 01. Available from: https://www.qatarday.com/News/japan-finds-another-moderna-vial-suspected-to-contain-foreign-substance/3285/0
32. Phillips J. White Floating Contaminants Found in Pfizer Vaccine in Several Japanese Cities: Officials. [cited 2021 Sept 23] Available from: https://www.theepochtimes.com/white-floating-contaminants-found-in-pfizer-vaccine-in-several-japanese-cities-officials_3999133.html?welcomeuser=1
33. RNZ. Covid-19 briefing: Spain supplies NZ with 250,000 extra vaccine doses; 2021 September 9. Available from:
https://www.rnz.co.nz/news/national/451121/covid-19-briefing-spain-supplies-nz-with-250-000-extra-vaccine-