Complaint to the Broadcasting Standards Authority (BSA)

NZDSOS has complained to the BSA about an appallingly one-sided broadcast by Seven Sharp about what is in the Pfizer Comirnaty injection.
Here is the original broadcast.
Our Complaint to the BSA
Thursday, 10 March 2022
NZDSOS Evidential response to TVNZ and BSA re Seven Sharp item, 13th of October 2021
As you are aware NZDSOS initiated a complaint with TVNZ in relation to the segment titled “What’s in the jab?” that aired on TVNZ’s Seven Sharp on the 13th of October 2021. After reviewing the contents of the segment, NZDSOS identified numerous breaches of the Broadcasting Standards Authority Free-To-Air Television Code Standard 9 – Accuracy. On the 7th of December we received a response from TVNZ which stated that no breach of standard 9 could be identified. We view their response as inadequate and refer the matter to the Broadcasting Standards Authority to pursue further investigation and review of their decision. TVNZ correctly re-iterated the concerns that we raised but failed to address them in their response.
Specifically, NZDSOS after reviewing the segment concluded that Dr Nikki Turner:
- Failed to accurately state the key components of the Pfizer Comirnaty Product.
- Repeatedly made misleading claims that Pfizer Comirnaty Product is “safe for everybody” (apart from her stated very small group of less than 100 people who may have severe allergic reactions to the PEG component of the Comirnaty product)
- Failed to indicate that the Pfizer Comirnaty product has been linked both locally and internationally to known serious side effects in adults and children including heart inflammation (myocarditis and pericarditis) heart attacks, strokes and death.
- Incorrectly stated that the Pfizer Comirnaty product was completely safe for pregnant women, while in truth there have not yet been any specific clinical safety trials during pregnancy to determine its safety parameters.
- Completely misleads the audience with her simplistic comments that totally ignore the known serious side effects which are clearly stated in the Pfizer Comirnaty product application to Medsafe.
The response on 7 Dec 2021 provided by TVNZ was sadly lacking in detail and failed to address the specific concerns raised by NZDSOS in their review of Dr Nikki Turner’s segment. The limited response provided by TVNZ includes:
- Dr Nikki Turner is a credible expert on the subject of immunisation so there was no reason for Seven Sharp to doubt the accuracy of her statements.
- Dr Nikki Turner summarised the ingredients of the vaccine, in necessarily simplified terms for viewers without specialist training.
- Dr Nikki Turner did not claim that the vaccine did not cause side effects. Her comments related to the proportion of people who are unable to get the vaccine and the level of risk for the vast majority of the population, both of which are low.
- The safety and efficacy of the vaccine has been well established. Medsafe states that “the protective benefits of vaccination against COVID-19 far outweigh the potential risks of vaccination”
In their non-substantive response TVNZ have essentially claimed the accuracy of their segment is ensured through their deferral to the expert opinion of Dr Nikki Turner. They also claim that the safety of the vaccines has been established by authorities such as the MOH, Medsafe and CDC. As these two points of deferral are the basis of their claim for accurate reporting the following response will focus on these.
1) Dr Nikki Turner is a credible expert on the subject of immunisation so there was no reason for Seven Sharp to doubt the accuracy of her statements.
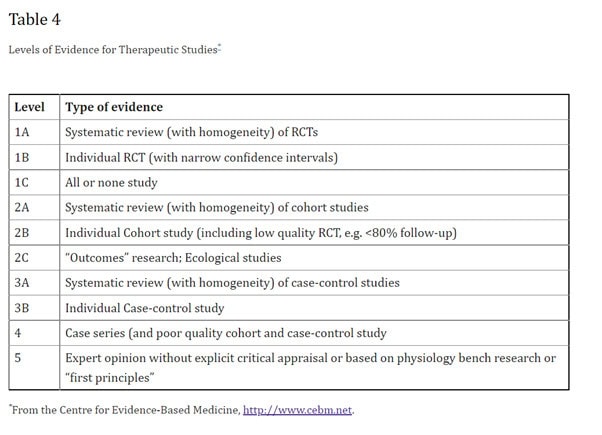
It is a sad indictment upon New Zealand Media organisations in general that they lack the capacity to be able to evaluate scientific data on their own merit and perpetually defer this responsibility to “experts”. Science and medicine prioritise evidence within a hierarchy, stratifying it in accordance with its relative strength. It is widely recognised that expert opinion is actually the lowest tier of evidence and that data derived from meta-analysis and randomised controlled trials form the upper tiers of this hierarchy.
It is even more concerning when these “experts” fail to stipulate in open declaration the point at which their opinions deviate from formalised scientific observation and enter into the realm of unsupported speculation. We state openly that in the absence of long-term clinical data there are no experts, there is only speculation. Science is built upon systematic controlled observation and not upon the intangible act of speculation. When there is a paucity of peer reviewed data available, medicine should operate on a precautionary principle which is underpinned by the maxim “let us first do no harm”.
In response to the notion that Dr Nikki Turner’s expertise in the field vaccination can be generalised to include this entirely new class of vaccines, we caution that on this occasion conventional methods of vaccination have been discarded in favour of the use of a novel gene transfer technology. The foundation of this newly adopted approach differs markedly from conventional vaccines, and the assumptions made by vaccinologists who are familiar with more conventional approaches to vaccination may well lack validity. The fundamental safety concerns relating to these products are at least in part, technology specific and may differ significantly from concerns arising from the use of conventional vaccines.
2) Dr Nikki Turner summarised the ingredients of the vaccine, in necessarily simplified terms for viewers without specialist training.
Dr Nikki Turner failed to provide an adequate description of the vaccine’s composition and mechanism of action describing the vaccine as being composed of ‘ little pieces of genetic material’ and ‘little fatty bubbles’. Consumers should have been advised that Pfizer Comirnaty mRNA vaccine available in New Zealand makes use of a new technology that uses a synthetic mRNA sequence delivered within protective lipid nanoparticle coating. There was no mention that both of these components are novel and there is no experience with their widespread use in healthy populations. There was no mention that the ‘little pieces of genetic material’ had been manipulated to enable them to persist in the human body (by substituting uridine for pseudo-uridine).
3) Dr Nikki Turner did not claim that the vaccine did not cause side effects. Her comments related to the proportion of people who are unable to get the vaccine and the level of risk for the vast majority of the population, both of which are low.
Accuracy of this broadcast is compromised by the omission of information that a qualified, registered, practicing medical doctor advising the public should include if true informed consent is sought from the population. Rather than upholding the ethical standards expected from a practicing doctor, this segment appears to be an advertorial/infomercial suggesting that the vaccine is entirely beneficial, suitable for everyone and there are no risks, alternatives or uncertainties.
In accordance with the Health and Disability Act 2000 and the MCNZ Statement on Informed Consent every consumer has the right to be fully informed when making a decision that could affect their future health. As a medical doctor it is Dr Nikki Turner’s obligation to provide an accurate description of the status of this vaccine and the level of certainty surrounding its safety.
See the next section for a more in-depth discussion of the absent information that should be provided by a licenced doctor.
4) The safety and efficacy of the vaccine has been well established. Medsafe states that “the protective benefits of vaccination against COVID-19 far outweigh the potential risks of vaccination”
The first sentence is opinion, rather than an accurate fact. Science evolves and new data is always coming to light and there should always be room to update knowledge and for discussion and debate. Studies of the safety and efficacy are ongoing. Referring to a statement from Medsafe does not mean it is accurate.
In an environment in which doctors are being demonised for providing full informed consent and acknowledging the deficiencies in the science supporting the broad deployment of mRNA vaccines, it is staggering that other doctors are openly misinforming the public by overstating their knowledge on the safety and efficacy of these vaccines with the full support of the media and government agencies.
Clinical Trials
Consumers have the right to be informed that the Pfizer Comirnaty mRNA vaccines that have been deployed in New Zealand are still involved in ongoing clinical trials and remain in the experimental phase until 2023. Medsafe’s initial provisional consent was granted on the basis of a clinical study citing only two months of clinical data. And while the relative risk reduction of 95% is often cited, the absolute risk reduction (effectiveness) is only 0.84%. Even if we accept the flawed use of relative risk reduction as it has been so vigorously reported by media outlets; the question remains, 95% effective at what exactly? In the correct parlance of evidence-based medicine the Pfizer BNT162b2 [mRNA] vaccinewas 95% effective at reducing mild to moderate symptoms in patients who were diagnosed with Covid-19 within a 2-month window. The study did not measure the ability of the vaccine to prevent hospitalisation or death, or its ability to reduce transmission. It is also incorrect to assume that these modest benefits would persist beyond the 2-month window over which the observations were made.
The data that has emerged since this early trial, confirm that the benefits of taking the mRNA vaccines are constrained by the rapid waning of the protective immune response and by the rapid emergence of new variants. Both of these phenomena place limits on the protective benefit of these vaccines relative to poorly defined risk that has been taken. The emphatic testimony of experts in the field has proven to be of little predictive value.
https://www.nejm.org/doi/full/10.1056/NEJMoa2034577?query=recirc_curatedRelated_article
https://www.nature.com/articles/d44151-022-00010-y
Provisional Consent
If the safety and efficacy have been well established, Dr Turner should have explained why the vaccine was only given provisional consent initially, and why provisional consent has been renewed without the progression to full consent. Consumers have not been advised that the provisional use authorisation granted by Medsafe for the Pfizer vaccine was subject to 58 conditions which were supposed to be addressed within specified time frames. Condition 5 is of particular concern as it asked that Pfizer “Provide data to further characterise the truncated and modified mRNA species present in the finished product” Medsafe was concerned that there were fragments of mRNA that may encode something other than the intact spike protein. The condition also raised the question of the potential for auto-immunity arising from immune interactions with the translated protein products of these uncharacterised mRNA fragments as the result of molecular mimicry. This is all information of which consumers have the right to be informed.
https://gazette.govt.nz/notice/id/2021-go338
Only One Possible Mechanism of Harm
Dr Nikki Turner indicated that the safety of the Pfizer Comirnaty vaccine is implicit because there is no virus in the vaccine. She failed consider that there are other mechanisms through which the vaccine might be harmful.
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7645850/
Safety Concerns in Scientific Literature
The injury rate associated with the deployment of Covid-19 vaccines is thirty times the rate reported with the more conventional Influenza vaccines. In addition, there have been over 1000 peer-reviewed publications internationally which describe the deficiencies in vaccine safety and adverse events reporting relating to the mRNA vaccines.
Important Potential Risk of ADE (Antibody-dependent enhancement) in Risk Management Plan (RMP) Summary
Instead of reassuring the public that this vaccine is completely safe and there are no concerns, Dr Turner as a practicing doctor, should have mentioned that in fact Medsafe was aware of a significant potential concern in addition to the anaphylaxis she did mention. This discussion should have addressed the identified serious potential risk of ADE. In this situation, vaccination actually worsens the clinical outcome of the patient instead of reducing the severity of the illness.
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7645850/
https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC7645850/
She should also have been aware that there was significant missing information and that this was relevant to the public discussion.
Safety in Immunocompromised People and Safety in People taking Medications.
In the segment in question, Dr Nikki Turner stated that the vaccine is perfectly safe for people taking medications and people with weakened immune systems. In contradiction to this, the Pfizer Comirnaty data sheet specifically states that these situations haven’t been studied.
See Medsafe datasheet: https://medsafe.govt.nz/Profs/datasheet/c/comirnatyinj.pdf
Immunocompromised individuals
The efficacy, safety and immunogenicity of COMIRNATY has not been assessed in immunocompromised individuals, including those receiving immunosuppressant therapy. The efficacy of COMIRNATY may be lower in immunosuppressed individuals.
The RMP Summary (linked above) specifically alerts to the fact that information about use in immunocompromised people is missing.
Table 5: Missing Information: Use in Immunocompromised Patients
Dr Turner states the vaccine is safe for people taking other medications. This is not what the advice document for doctors (the Medsafe datasheet) states.
4.5 Interactions with other medicines and other forms of interactions
No interaction studies have been performed. Concomitant administration of COMIRNATY with other vaccines has not been studied.
Safety During Pregnancy and Lactation
Dr Nikki Turner went on to claim that the vaccines are safe for women during pregnancy and while breast feeding, stating “We’re sure it’s a really good thing to breastfeed your babies, and to be vaccinated while you are breastfeeding.” The Pfizer Comirnaty data sheet cautions that there is limited experience with Comirnaty in pregnant women and cites limited animal study data. The data sheet goes on to confirm that they have not conducted studies to determine whether BNT162b2 [mRNA] is secreted in human breast milk or to determine the risks associated with this.
Pregnancy
There is limited experience with use of COMIRNATY in pregnant women. Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryo/fetal development, parturition or post-natal development (see Section 4.6 Fertility, pregnancy and lactation, Fertility). Administration of COMIRNATY in pregnancy should only be considered when the potential benefits outweigh any potential risks for the mother and fetus.
Lactation
It is unknown whether BNT162b2 [mRNA] is excreted in human milk. A combined fertility and developmental toxicity study in rats did not show harmful effects on offspring development before weaning (see Section 4.6 Fertility, pregnancy and lactation, Fertility).
The RMP Summary specifically alerts to the fact that information about use in pregnancy and during breastfeeding is missing.
Severity of Disease and therefore necessity of the Vaccine
Dr Nikki Turner took the position that the risks associated with infection with SARS-Cov-2 outweigh the risks associated with vaccination. The survival rates even for elderly with co-morbidities is still in the vicinity of >95% and the global infection fatality rate (IFR) is approximately 0.15% making the fatality rate associated with Covid-19 similar to influenza. It is also not possible to make an informed calculation of the comparative risks when long-term safety data is not available.
https://onlinelibrary.wiley.com/doi/epdf/10.1111/eci.13554
Being able to make a comparative assessment would also require that the totality of adverse event reports to CARM had been fully assessed.
Interference with DNA
While it seems that the media have relegated concerns about retro-integration (permanent alteration of the genome) to the realm of conspiracy theory, no such certainty exists in scientific circles amongst the people who have dedicated their careers to studying such questions. In fact, data from a paper published in late 2020 indicates that SARS-Cov-2 specific RNA sequences can be reverse transcribed and integrated into the human genome.
https://www.biorxiv.org/content/10.1101/2020.12.12.422516v1
Another more recent study has provided direct evidence of the reverse transcription and integration of Pfizer BNT162b2 [mRNA] into the DNA of a hepatic cell line. While the clinical significance of this is yet to be determined, this is direct evidence of an event that certain “experts” assured us, could not happen.
https://www.mdpi.com/1467-3045/44/3/73/htm
There is also emerging evidence which shows that the spike protein can migrate into the nucleus of human cells and can directly interfere with the mechanisms of DNA repair. This has raised concerns about the ability of the spike protein to impair adaptive immunity and act as a mutagen.
https://www.mdpi.com/1999-4915/13/10/2056/htm
Conclusion
As the interview segment shown by Seven Sharp was pre-recorded it is not possible for NZDSOS to determine whether edits have been made to the information provided by Dr Nikki Turner. We accept that the message delivered to the public may not be an accurate portrayal of the message that was intended to be delivered by Dr Nikki Turner. But as our complaint is based on the accuracy of the information provided, we can only contend with the information provided in the segment as it was shown on air. TVNZ also seem to have taken the position that in making reference to government and international agencies such as the Ministry of Health, Medsafe and CDC websites due diligence is implied. As you will see from the response above none of these sources are considered to be high level evidence within the hierarchy of evidence, as little peer reviewed scientific evidence is provided in support their policies. We refer the matter back to the broadcasting standards authority in the hope of achieving an objective and unbiased investigation of our concerns.
Kind Regards,
NZDSOS